DPDP Act Full Form And
Why It Changes Doctor Marketing Forever

The full form of DPDP is Digital Personal Data Protection Act. On the surface, this may sound like another regulatory acronym added to the long list pharma teams already deal with.
In reality, DPDP represents one of the most consequential shifts doctor marketing has seen in decades.
Unlike earlier guidelines or sector-specific advisories, DPDP fundamentally redefines how doctor data can be collected, stored, shared, and used. It does not simply add a layer of compliance. It changes the assumptions on which doctor engagement models were built.
For pharma marketing teams, understanding the DPDP Act full form is only the starting point. What matters far more is understanding how this law permanently alters doctor databases, consent logic, CRM usage, omnichannel outreach, and accountability for marketing execution.
This article explains why DPDP changes doctor marketing forever, not temporarily, and why going back to old models is no longer an option.
What DPDP Actually Stands For in Practice
Digital Personal Data Protection Act is the formal expansion of DPDP. But each word in that name carries practical consequences for pharma marketing.
Digital means the law applies to data processed in digital form. This includes CRM systems, marketing automation platforms, WhatsApp campaigns, email tools, data warehouses, AI models, and analytics systems.
Personal data refers to any data that can identify an individual. In pharma marketing, this clearly includes doctor names, phone numbers, email addresses, clinic locations, engagement history, digital identifiers, and in many cases inferred preferences.
Protection is not limited to storage security. It includes how data is collected, why it is collected, how it is used, who it is shared with, and when it must be deleted.
Act means this is enforceable law, not guidance. The expectations apply regardless of intent, scale, or legacy practices.
When combined, DPDP does not just regulate data handling. It reshapes how doctor marketing must be designed from the ground up.
Why Doctor Marketing Is Directly in DPDP’s Scope
There is a persistent misconception that DPDP primarily targets consumer businesses or large technology platforms. This belief has led some pharma teams to underestimate its relevance.
Doctor marketing sits squarely within DPDP’s scope because doctors are identifiable individuals and their data is processed digitally for commercial purposes. Whether the intent is scientific education, brand communication, or engagement analytics, the underlying data is personal data under the law.
Even if the data is professionally oriented, it does not lose its personal nature. DPDP does not distinguish between personal and professional identities in the way older data practices often assumed.
As a result, traditional justifications such as “this is B2B data” or “this is industry practice” no longer hold legal weight.
How Doctor Marketing Worked Before DPDP
To understand why DPDP changes everything, it is important to acknowledge how doctor marketing typically evolved over time.
Doctor data was aggregated from conferences, third-party vendors, field force interactions, online portals, and partner ecosystems. Databases grew over years, often without clear records of original consent or purpose.
Marketing teams focused on reach, frequency, and segmentation. Consent, when present, was usually broad and static. Once captured, it was rarely revisited. Campaign execution prioritised speed and scale.
This model functioned largely because regulatory scrutiny around data usage was limited and enforcement was minimal.
DPDP dismantles this foundation.
Why the DPDP Act Full Form Signals a Permanent Shift
DPDP is not a transitional regulation that can be waited out. It represents India’s alignment with global data protection thinking while introducing its own accountability structure.
What makes DPDP particularly disruptive for doctor marketing is that it does not allow grandfathering of non-compliant data. Existing databases are not automatically legitimised. Legacy practices are not protected by historical norms.
This means pharma companies must actively reassess what data they hold, why they hold it, and whether they are entitled to use it in the way they currently do.
This shift is permanent because it is embedded into how data rights are defined and enforced going forward.
Consent Under DPDP Changes the Logic of Doctor Engagement
Consent is the single most misunderstood aspect of DPDP in pharma marketing.
Earlier models treated consent as a one-time gateway. Once a doctor agreed to receive communication, their data flowed freely across channels and campaigns.
DPDP replaces this with a more granular and dynamic model. Consent must be explicit, informed, purpose-specific, and revocable. This means consent is no longer a static attribute of a doctor record. It is a live condition that governs every interaction.
For example, consent to receive scientific updates does not automatically extend to promotional messages. Consent for email does not automatically cover WhatsApp. Consent given two years ago may no longer be valid if the purpose has evolved.
This fundamentally alters how campaigns must be planned and executed.
Why CRM Systems Alone Are No Longer Enough
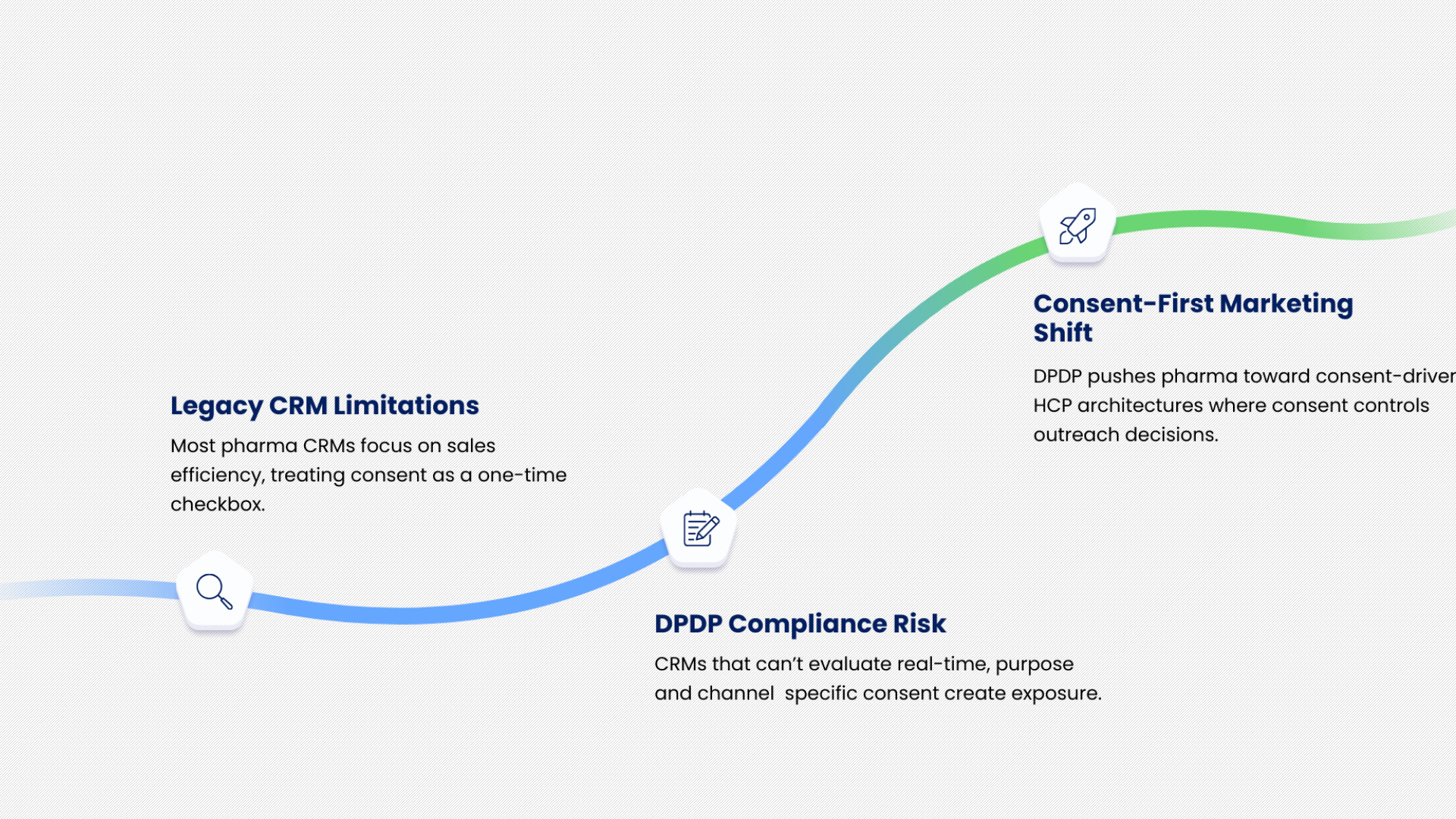
Many pharma CRMs were designed to optimise sales force productivity and campaign reach. Consent, if captured, was often treated as a compliance checkbox rather than an operational control. DPDP exposes this limitation.
A CRM that cannot dynamically evaluate consent at the moment of outreach creates risk.
A CRM that cannot map consent to specific purposes or channels cannot support DPDP-compliant execution. This is why DPDP pushes pharma marketing toward consent-first HCP marketing architectures, where consent intelligence is embedded into campaign decisioning rather than layered on top.
Omnichannel Doctor Marketing After DPDP
Omnichannel engagement has been a major focus for pharma marketing over the past decade. DPDP does not eliminate omnichannel strategies, but it enforces discipline in how they are applied.
Each channel now requires explicit permission. Each campaign requires purpose alignment. Each interaction must be defensible.
This means that blasting the same message across email, WhatsApp, and digital ads without channel-specific consent is no longer viable. Campaigns must be narrower, more deliberate, and better governed.
While this may reduce superficial reach, it significantly improves quality and sustainability of engagement.
Agencies, Vendors, and Shared Responsibility Myths
Another area where DPDP changes long-standing assumptions is agency accountability.
In many organisations, marketing execution is heavily outsourced. Agencies run campaigns, manage data flows, and operate marketing tools. This has led to a belief that agencies absorb compliance risk.
DPDP does not support this belief.
The data fiduciary, usually the pharma company, remains accountable for how personal data is processed. Agencies become data processors operating under defined instructions.
This means pharma companies must take greater ownership of how agencies handle doctor data, including consent verification, data sharing, and audit readiness.
DPDP and AI Driven Doctor Marketing
AI is increasingly used to personalise content, recommend next best actions, and optimise engagement timing. These capabilities rely heavily on personal data and behavioural signals.
DPDP does not ban AI usage. But it raises expectations around data provenance, purpose limitation, and consent propagation.
If a doctor withdraws consent, that withdrawal must reflect across CRM systems, marketing tools, and AI models. If data was collected for one purpose, AI outputs cannot quietly extend it into another.
This pushes pharma teams to think about AI governance as part of marketing design, not just technology deployment.
Why DPDP Forces a Rethink of Marketing Metrics
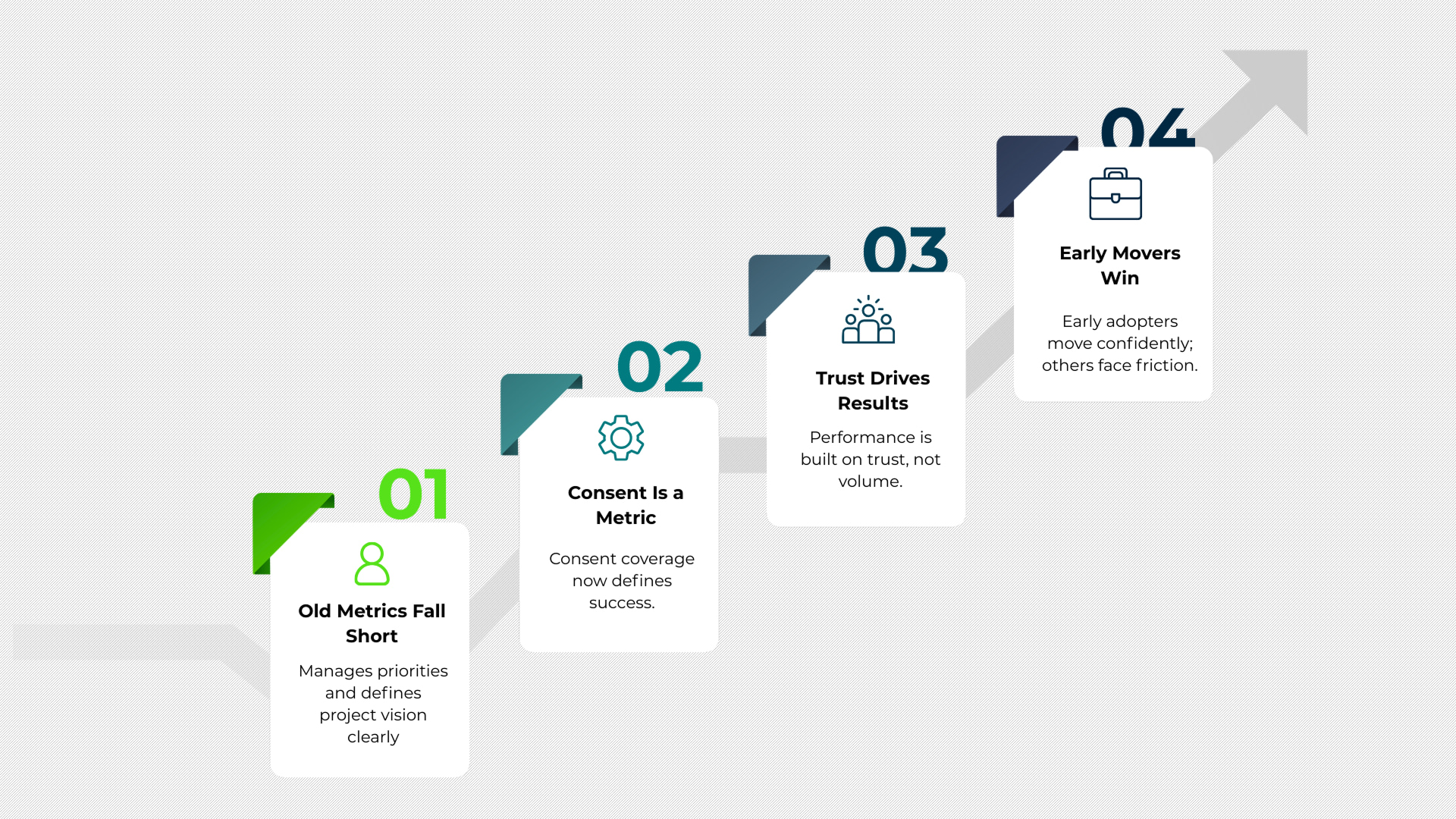
Traditional marketing metrics focused on reach, opens, clicks, and frequency. DPDP introduces a new dimension that cannot be ignored.
Consent coverage becomes a critical metric. Auditability becomes a requirement. Data lineage matters as much as campaign performance.
This shifts success from volume driven metrics to trust driven execution.
Teams that adapt early will find it easier to operate confidently. Teams that delay will face increasing friction as audits, partner scrutiny, and regulatory expectations rise.
What Pharma Marketing Leaders Should Do Now
The most effective response to DPDP is proactive redesign, not reactive compliance.
Marketing leaders should start by mapping all doctor data sources and understanding consent gaps. They should evaluate whether their current tools can support purpose based consent enforcement. They should align agencies, legal teams, and IT around shared accountability.
Most importantly, they should recognise that DPDP is not a temporary hurdle. It is the new operating environment.
Why DPDP Changes Doctor Marketing Forever
DPDP permanently changes doctor marketing because it alters the power balance around data. Doctors gain clearer rights. Pharma companies gain clearer responsibilities. Systems and workflows must adapt.
This is not about avoiding penalties. It is about building marketing operations that can scale without hidden risk.
Teams that continue to rely on legacy practices will find those practices increasingly fragile. Teams that invest in compliant foundations will gain long-term stability.
Frequently Asked Questions on Purpose Limitation Under DPDP
Closing Perspective and CTA
Understanding the DPDP Act full form is only the first step. The real challenge lies in translating that understanding into daily marketing execution.
Doctor engagement under DPDP requires consent first thinking, stronger data governance, and marketing systems designed for accountability.
If you are evaluating how to run doctor engagement and HCP marketing under DPDP without disrupting growth, this page outlines how DPDP-compliant HCP marketing is being implemented in practice.
