Consent Withdrawal Under DPDP:
Why Permission Changes Must Cascade Across Systems ?

Consent withdrawal is one of the most powerful rights granted to individuals under the Digital Personal Data Protection Act 2023. For pharma companies, it is also one of the most operationally misunderstood obligations.
Many organisations believe that consent withdrawal is handled once a doctor unsubscribes from an email or opts out of a WhatsApp message. Under DPDP, this belief is dangerously incomplete.
Consent withdrawal is not a channel level event. It is a system wide instruction that must cascade across every platform, dataset, workflow, and vendor that processes the individual’s data for that purpose.
This article explains how consent withdrawal should cascade across pharma systems, why partial handling creates compliance exposure, and what operational changes are required to meet DPDP expectations at scale.
What Consent Withdrawal Means Under DPDP ?
Under DPDP, consent withdrawal means the individual revokes permission for their personal data to be processed for a specific purpose.
Once consent is withdrawn, the organisation must stop processing the data for that purpose immediately. There is no grace period and no partial compliance.
Processing includes outreach, analytics, profiling, and any automated decision making that relies on the data.
This makes consent withdrawal a live operational trigger, not a documentation update.
Why Pharma Commonly Mismanages Consent Withdrawal ?
 Pharma systems evolved in silos.
Pharma systems evolved in silos.
Email tools handle unsubscribes. WhatsApp platforms handle opt outs. CRMs store flags. Analytics tools continue to process historical data. Vendors retain copies.
These systems rarely communicate effectively.
As a result, consent withdrawal is handled inconsistently. Outreach may stop in one channel but continue in another. Analytics may still process data long after consent is withdrawn.
Under DPDP, this fragmentation is non compliant.
Consent Withdrawal Must Be Purpose Specific
Consent withdrawal applies to a specific purpose.
A doctor may withdraw consent for promotional communication but continue to allow educational updates. A patient may withdraw consent for marketing but remain enrolled in a support program.
Systems must respect this granularity.
Treating withdrawal as a blanket removal or ignoring purpose distinctions creates both compliance and engagement problems.
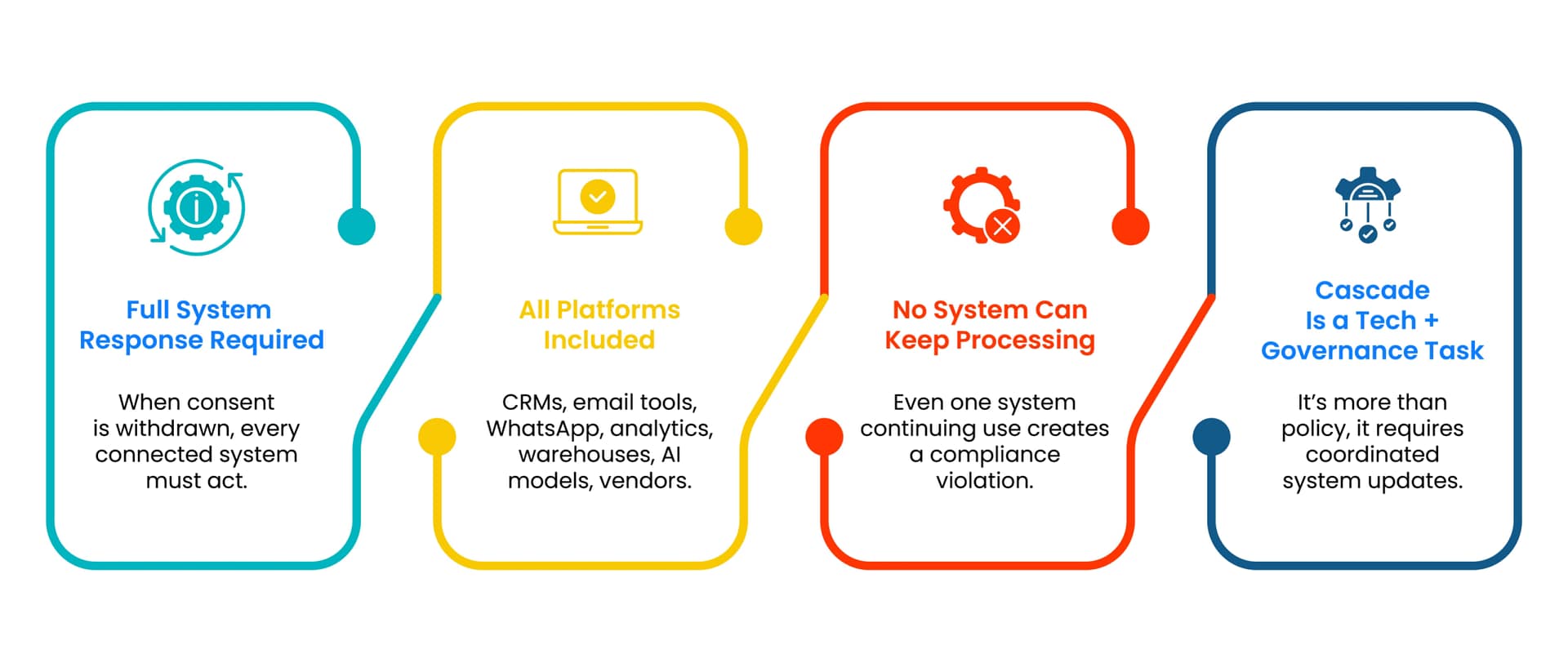
Cascade Means Every System Must Respond
When consent is withdrawn, every system that processes the relevant data must respond.
This includes CRMs, email platforms, WhatsApp systems, analytics tools, data warehouses, AI models, and vendor systems.
If any system continues processing the data for the withdrawn purpose, the organisation remains in violation.
Cascade is therefore a technical and governance challenge, not a policy statement.
Why Channel Level Opt Outs Are Insufficient ?
 Channel level opt outs address only one symptom.
Channel level opt outs address only one symptom.
Unsubscribing from email stops email delivery but does not stop WhatsApp messaging, ad targeting, analytics processing, or data sharing.
DPDP does not recognise channel level compliance as sufficient if processing continues elsewhere.
Consent withdrawal must be enforced across all channels and uses for that purpose.
The Role of a Central Consent Authority
To cascade consent withdrawal effectively, pharma organisations need a central consent authority.
This authority maintains the definitive state of consent by individual, purpose, and channel. All systems must query this authority before processing data.
When consent changes, the authority updates immediately and downstream systems must respond.
Without this centralisation, cascade is impossible to guarantee.
This is where DPDP-compliant HCP marketing architectures become essential. They provide a single source of truth for consent and enforce it across execution layers.
Real Time Versus Batch Propagation
Timing matters.
Consent withdrawal must be propagated in real time or near real time. Batch updates that run daily or weekly create windows of non compliance.
If a doctor withdraws consent at 10 AM and receives a message at noon, the organisation is exposed.
Real time propagation requires event driven integration rather than scheduled syncs.
Handling Consent Withdrawal in CRMs
CRMs are often the first system updated.
However, updating a flag in the CRM is not enough. CRMs must notify all integrated systems immediately.
CRMs should not be treated as passive storage. They must participate in the cascade.
If the CRM cannot propagate changes instantly, it cannot be relied upon as the sole consent system.
Handling Consent Withdrawal in Email Platforms
Email platforms typically handle unsubscribes well within their own environment.
The problem arises when unsubscribes do not update central consent records or other systems.
Email opt outs must trigger updates to the central consent authority. They must also prevent future list exports or campaign targeting that bypasses unsubscribe lists.
Email platforms should not be treated as isolated compliance silos.
Handling Consent Withdrawal in WhatsApp Systems
WhatsApp opt outs are often handled through keywords or platform level settings.
These opt outs must be captured centrally and enforced across all WhatsApp templates, campaigns, and vendors.
Manual handling or delayed updates increase risk significantly.
At scale, WhatsApp consent withdrawal must be automated end to end.
Consent Withdrawal and Digital Advertising
Digital advertising presents one of the hardest cascade challenges.
Audience lists may exist on external platforms. Retargeting pixels may continue collecting data. Campaigns may continue delivering ads automatically.
Consent withdrawal must trigger removal from audience lists and stop further targeting.
Pharma companies must design integrations that allow rapid suppression of withdrawn individuals from ad platforms.
Analytics and Data Processing After Withdrawal
Consent withdrawal applies not only to outreach but also to analytics.
Processing personal data for analytics after withdrawal is non compliant if analytics was part of the withdrawn purpose.
Systems must stop using the data and consider deletion or anonymisation. Ignoring analytics processing is a common oversight.
AI Models and Consent Withdrawal
AI systems often rely on historical data.
If consent is withdrawn, organisations must evaluate whether continued use of that data in models is allowed.
In many cases, data must be excluded from future processing. This may require retraining models or isolating withdrawn data.
DPDP forces organisations to confront AI lifecycle governance.
Vendor Systems and Consent Cascade
Vendor systems are often the weakest link.
Agencies and technology partners may hold copies of data. Consent withdrawal must be communicated to them and enforced.
Vendor contracts should include obligations for immediate compliance with consent changes. Without vendor integration, cascade is incomplete.
Auditing Consent Withdrawal Cascades
Auditors will test consent withdrawal.
They may submit a withdrawal request and observe whether messages continue. They may inspect system logs to see how updates propagate.
Organisations must be able to demonstrate that withdrawal triggers a cascade across systems. Partial compliance will not pass scrutiny.
Designing Consent Withdrawal as an Event
The key design shift is treating consent withdrawal as an event, not a flag. Events trigger workflows. Flags do not.
An event driven approach ensures that every system reacts immediately and consistently. This design supports scale and reduces reliance on manual intervention.
Operational Ownership of Consent Cascades
Consent cascade requires clear ownership.
Someone must own the consent authority. Someone must own system integrations. Someone must monitor failures.
Treating consent withdrawal as everyone’s responsibility often results in no one owning it. Clear accountability is essential.
Business Impact of Proper Consent Cascade
While implementing full cascade is complex, it delivers benefits.
Complaints decrease. Trust improves. Audit readiness strengthens. Risk reduces. Operational clarity increases because systems behave predictably.
Frequently Asked Questions on Consent Withdrawal Cascades
Closing Perspective and CTA
Consent withdrawal under DPDP is not a minor operational detail. It is a system wide instruction that must be enforced consistently and immediately.
Pharma companies that treat withdrawal as a channel level opt out expose themselves to serious compliance risk.
Those that design event driven, system wide cascades will be able to operate confidently under DPDP.
If you are evaluating how to implement DPDP-compliant HCP marketing with full consent withdrawal cascade across systems, this page explains how consent-first architectures are being implemented in real pharma environments.
