Are Pharma Companies Data Fiduciaries Under DPDP Act?

One of the most important and misunderstood questions under the Digital Personal Data Protection Act 2023 is whether pharmaceutical companies qualify as data fiduciaries. Many pharma organisations still assume that data responsibility sits primarily with technology vendors, CRM providers, digital agencies, or data partners.
The DPDP Act does not support this assumption.
In most real-world scenarios, pharma companies clearly meet the definition of a data fiduciary under the law. This classification carries direct legal and operational consequences for marketing, commercial, medical, and digital teams.
This article explains what it means to be a data fiduciary under DPDP, why pharma companies almost always fall into this category, and how this designation reshapes accountability across doctor engagement and marketing workflows.
What Does Data Fiduciary Mean Under DPDP Act?
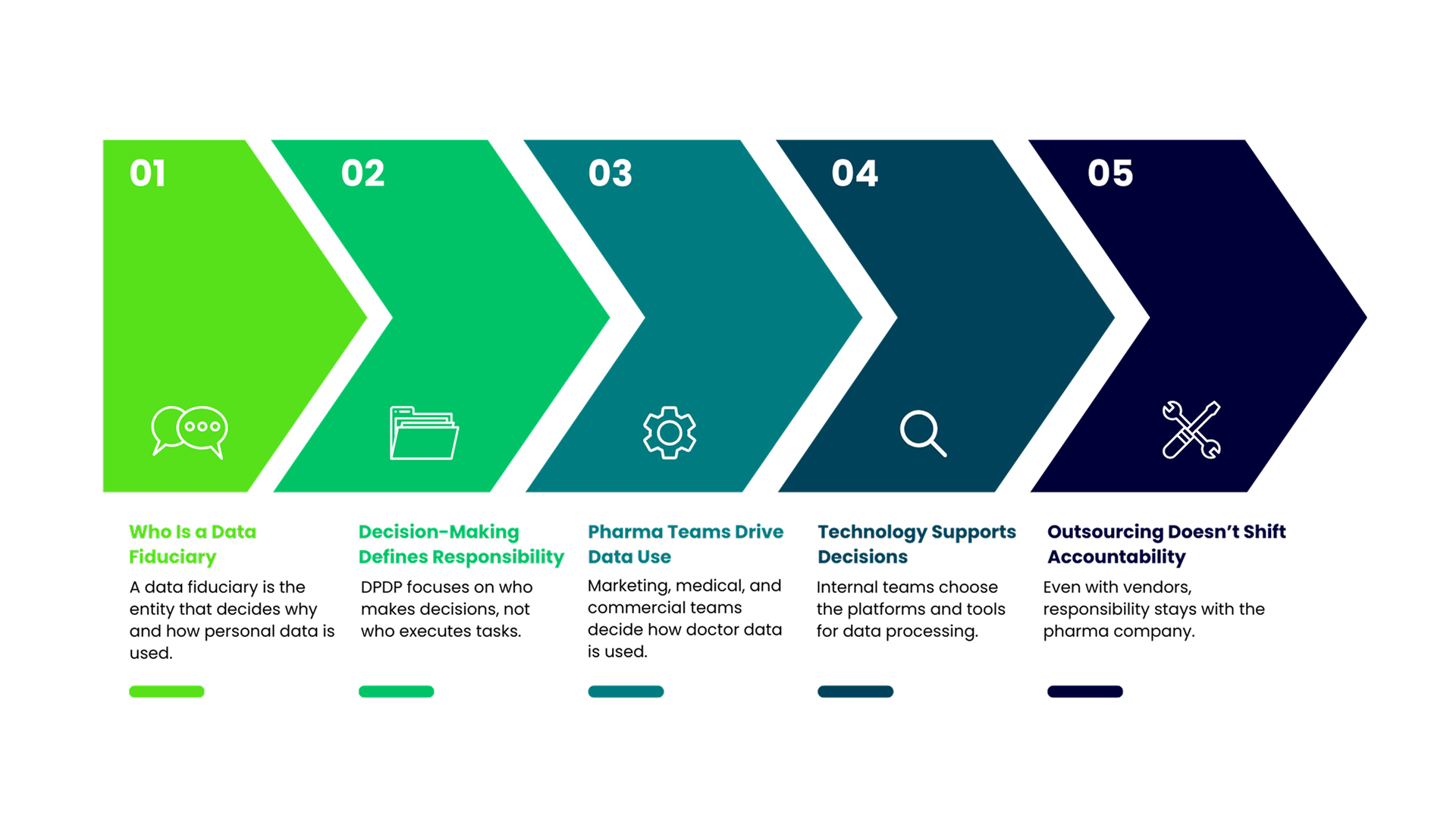
Under the DPDP Act, a data fiduciary is the entity that determines why and how personal data is processed.
This definition is intentionally broad. It focuses on decision-making power rather than technical execution. The entity that decides the purpose of data use and the means of processing is the one held accountable.
In pharma organisations, these decisions are typically made by internal teams. Marketing decides which doctors to target. Medical teams decide what content is shared. Commercial teams define engagement strategies. Technology teams select platforms and tools.
Even when execution is outsourced, the underlying decisions originate within the pharma company.
Why Pharma Companies Almost Always Qualify as Data Fiduciaries ?
Pharma companies control the core elements that define data fiduciary status.
They decide which personal data is collected, whether it relates to doctors, patients, or other healthcare professionals. They define the purpose of data use, such as marketing communication, medical education, or engagement analytics. They choose the channels through which communication occurs. They determine how long data is retained and when it is deleted.
Vendors and agencies may process data on behalf of pharma companies, but they do not typically decide the purpose or scope of processing independently.
This places pharma companies firmly in the role of data fiduciary under DPDP.
Common Misconception: Agencies as Data Fiduciaries
A frequent misconception in pharma marketing is that agencies or platform providers act as data fiduciaries because they operate the tools and run the campaigns.
DPDP draws a clear distinction between data fiduciaries and data processors. Data processors act on instructions provided by the data fiduciary. They do not define the purpose of processing.
In most pharma marketing arrangements, agencies follow briefs, campaign plans, and data access rules defined by the pharma company. This makes them data processors, not data fiduciaries.
The responsibility for compliance remains with the pharma company.
How Data Fiduciary Status Affects Pharma Marketing Teams
Being classified as a data fiduciary has practical consequences for marketing and commercial teams.
Marketing teams must ensure that data used in campaigns is collected lawfully and with valid consent. They must ensure that data is used only for the purposes communicated to doctors. They must be able to demonstrate compliance during audits or investigations.
This shifts compliance from being a background legal concern to an operational responsibility embedded in daily workflows.
Data Fiduciary Responsibilities in Doctor Engagement
Doctor engagement relies heavily on personal data. Contact details, engagement history, preferences, and digital interaction data all qualify as personal data under DPDP.
As data fiduciaries, pharma companies must ensure that this data is accurate, up to date, and used appropriately. They must respect consent withdrawal and provide mechanisms for doctors to exercise their data rights.
This directly affects how doctor databases are managed and how engagement programs are designed.
Impact on CRM Systems and Marketing Platforms
CRM systems play a central role in doctor engagement. However, many CRMs were designed for sales enablement rather than regulatory accountability.
Under DPDP, data fiduciaries must ensure that CRMs support consent tracking, purpose mapping, and enforcement at the point of execution. A CRM that allows outreach without validating consent introduces compliance risk.
This is why DPDP-compliant HCP marketing architectures are becoming critical for pharma organisations. They connect data fiduciary responsibilities directly with marketing execution systems.
Vendor Management Under Data Fiduciary Obligations
Being a data fiduciary also affects how pharma companies work with vendors.
Contracts must clearly define roles and responsibilities. Data access must be limited to what is necessary. Vendors must follow documented instructions. Audit rights and safeguards must be in place.
Pharma companies cannot rely on vendor assurances alone. They must actively govern how personal data is handled across the ecosystem.
Data Fiduciary Role in AI Driven Marketing
AI driven marketing introduces additional complexity.
AI systems often rely on large datasets and behavioural signals. As data fiduciaries, pharma companies must ensure that AI models are trained on lawfully collected data and that outputs respect consent scope and purpose limitation.
Consent withdrawal must propagate across AI systems, not remain confined to source databases. This requires closer integration between data governance and AI deployment.
What Happens When Pharma Companies Ignore Fiduciary Status ?
 Ignoring data fiduciary responsibilities does not eliminate liability. It increases risk.
Ignoring data fiduciary responsibilities does not eliminate liability. It increases risk.
If a violation occurs, regulators will look at who decided why the data was used. In most cases, this points back to the pharma company, not the vendor.
This can lead to penalties, audits, and reputational damage. It can also disrupt marketing operations if data usage is restricted suddenly.
Preparing Pharma Teams for Data Fiduciary Accountability
The most effective response is proactive preparation.
Pharma companies should clearly document data purposes, consent mechanisms, and processing workflows. Marketing teams should be trained to understand fiduciary responsibilities. Systems should be evaluated for consent enforcement capabilities.
Data fiduciary accountability should be treated as a design principle rather than an afterthought.
Why Data Fiduciary Status Is Not Optional ?
Under DPDP, data fiduciary status is not something a company can opt into or out of. It is determined by how data decisions are made in practice.
For pharma companies, the conclusion is clear. If you decide how doctor or patient data is used, you are a data fiduciary.
Accepting this reality is the first step toward compliant and sustainable marketing operations.
Frequently Asked Questions on Data Fiduciary Status in Pharma
Closing Perspective and CTA
The DPDP Act makes one thing clear. Control over data decisions brings accountability.
For pharma companies, data fiduciary status is not a legal technicality. It is a practical reality that shapes how doctor engagement, marketing execution, and digital transformation must operate.
If you are evaluating how to meet data fiduciary responsibilities while running DPDP-compliant HCP marketing, this page explains how consent-first, accountable engagement models are being implemented in real pharma environments.
