Why Pre-DPDP Consent Models No Longer Work in Pharma

For years, consent in pharma marketing was treated as a procedural formality rather than a living operational requirement. Once a doctor agreed to receive communication, that approval was assumed to apply indefinitely and across channels. Databases grew, campaigns scaled, and consent records remained largely unchanged in the background.
The Digital Personal Data Protection Act 2023 changes this assumption completely.
Pre-DPDP consent models were built for a different regulatory environment. They prioritised reach and speed over traceability and purpose control. Under DPDP, these models are no longer sufficient, and in many cases, they expose pharma companies to real compliance and operational risk.
This article explains why traditional consent approaches fail under DPDP, what specifically breaks in existing pharma workflows, and how marketing teams must rethink consent as part of execution rather than documentation.
How Consent Was Traditionally Handled in Pharma Marketing
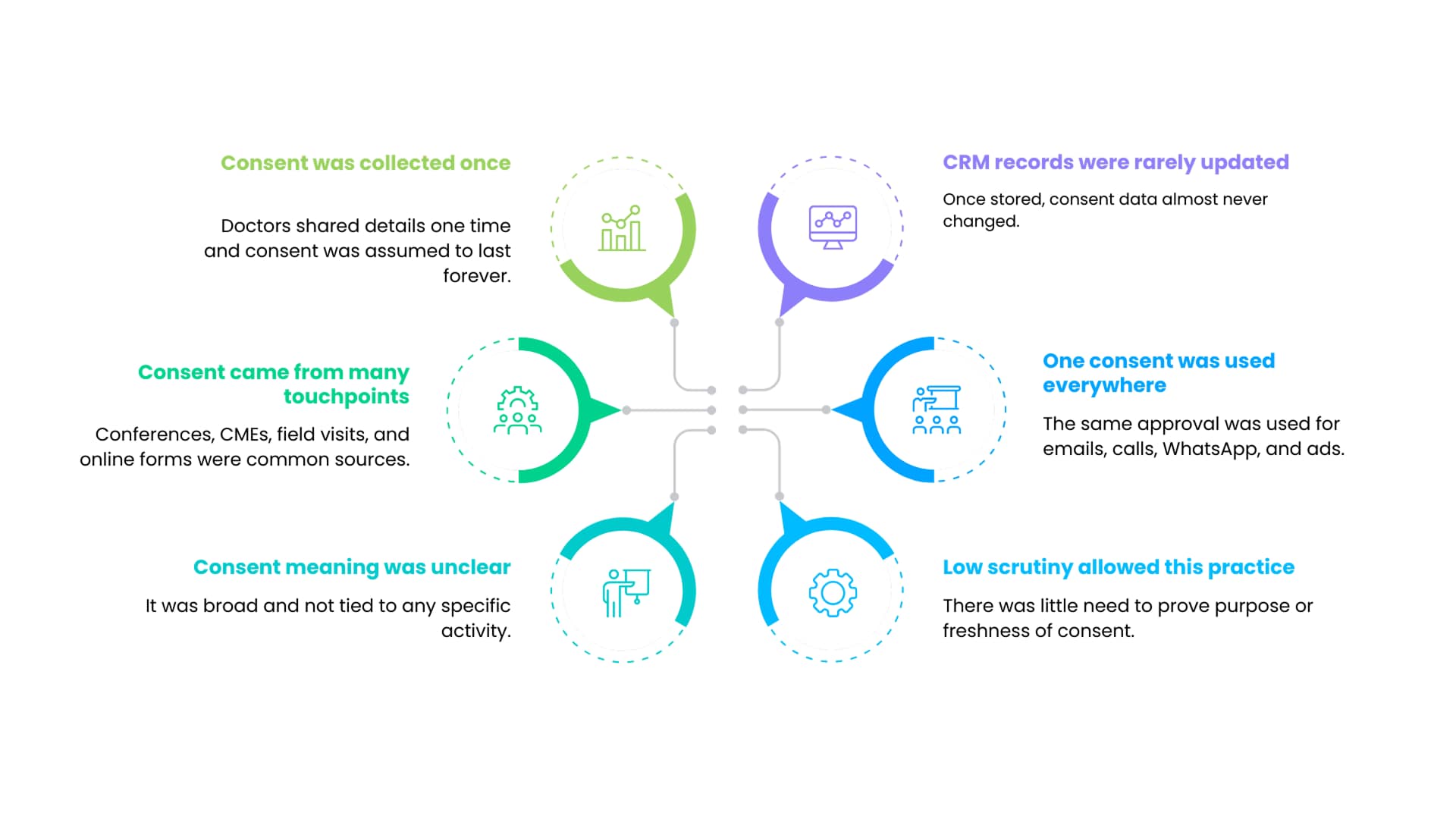
Historically, consent in pharma marketing was often captured once and reused many times. Doctors provided contact details during conferences, CME programs, field force interactions, or online registrations. That consent was usually broad and loosely defined.
Once stored in a CRM, the consent record rarely changed. It was assumed to apply to email, phone calls, WhatsApp messages, digital ads, and future campaigns that had not yet been designed.
This model worked largely because regulatory scrutiny around digital data use was limited. There was little requirement to prove purpose alignment or consent freshness.
DPDP invalidates this approach.
What DPDP Changes About Consent Fundamentally
DPDP introduces a stricter definition of valid consent. Consent must now be explicit, informed, specific to a purpose, and capable of being withdrawn easily.
This changes the nature of consent from a static record to a dynamic control mechanism. Consent is no longer something captured at onboarding and forgotten. It becomes a condition that governs every instance of data use.
For pharma marketing teams, this means consent must be checked at the point of execution, not just at the point of collection.
Why Generic Opt-In Models Fail Under DPDP
One of the most common legacy practices is the generic opt-in. A doctor agrees to receive communication from a pharma company, often without clarity on channel, content type, or duration.
Under DPDP, such opt-ins are insufficient because they do not define purpose clearly. Consent for scientific updates does not automatically extend to promotional messaging. Consent for email does not automatically cover WhatsApp or SMS.
Without purpose clarity, pharma companies cannot demonstrate lawful use of data if challenged.
This is why pre-DPDP opt-in models no longer hold up under regulatory scrutiny.
Channel Blind Consent Is No Longer Defensible
Earlier consent models rarely distinguished between communication channels. Once a doctor’s contact details were available, teams assumed they could be used across platforms.
DPDP requires channel specific consent. A doctor may be comfortable receiving email communication but not instant messaging. They may agree to digital content but not targeted advertising.
Marketing systems that do not differentiate consent by channel risk violating DPDP requirements during execution.
Consent Validity Over Time
Another weakness of pre-DPDP consent models is the assumption of permanence. Consent captured years ago is often treated as valid indefinitely.
DPDP challenges this assumption. Consent must remain relevant to the stated purpose. If the purpose evolves or expands, fresh consent may be required. If consent is withdrawn, processing must stop immediately.
This introduces the concept of consent freshness and lifecycle management, which most legacy systems do not support.
Impact on CRM and Campaign Automation
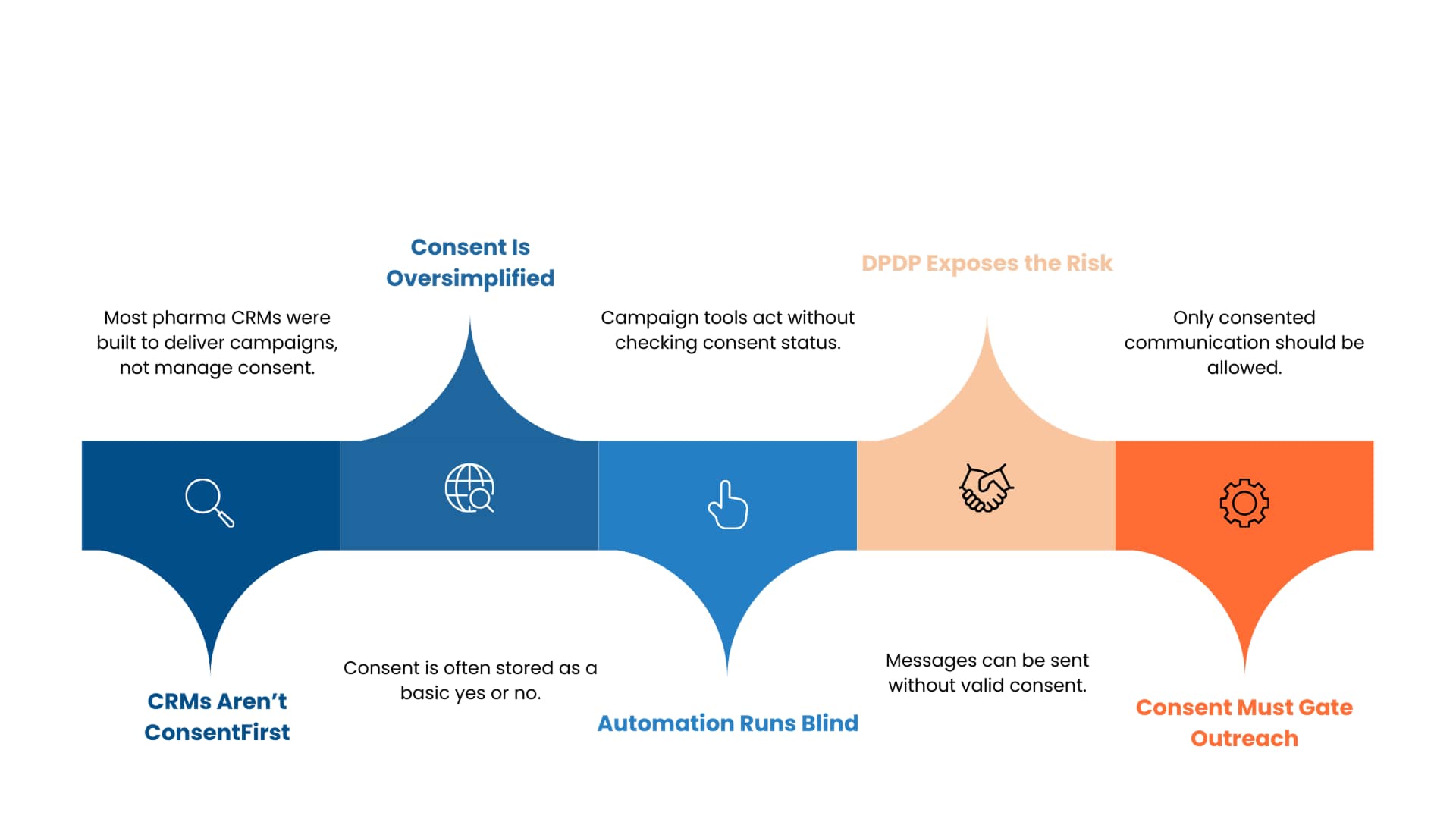
Most pharma CRMs were designed to optimise segmentation and campaign delivery, not to enforce consent logic dynamically.
Consent is often stored as a simple yes or no field, without purpose mapping or withdrawal workflows. Campaign automation tools then operate independently of consent context.
Under DPDP, this separation creates risk. Marketing automation must be consent aware. Outreach must be blocked automatically where consent does not exist.
This is where DPDP-compliant HCP marketing architectures become essential, because they integrate consent enforcement directly into campaign execution.
Agencies and the Illusion of Delegated Consent Responsibility
In many pharma organisations, agencies manage campaign execution and sometimes even data handling. This has created an illusion that agencies carry consent responsibility.
DPDP does not support this view. The data fiduciary remains responsible for ensuring that consent is valid and enforced, regardless of who executes the campaign.
Pre-DPDP consent models often fail because they rely on agency processes that are not designed for regulatory accountability.
Why Pre-DPDP Consent Breaks During Audits
Audits expose the weaknesses of legacy consent models.
When asked to demonstrate how consent was obtained, for what purpose, and how it was enforced across systems, many teams struggle. Records are fragmented. Purpose definitions are vague. Withdrawal processes are manual or non-existent.
DPDP raises expectations around auditability. Companies must be able to show not just that consent exists, but how it governs actual data use.
Consent and AI Driven Marketing
AI driven marketing adds another layer of complexity.
AI models trained on historical data may continue to use signals derived from consent that is no longer valid. Without proper consent propagation, withdrawal requests may not reach downstream systems.
Pre-DPDP consent models do not account for these complexities, making them unsuitable for AI enabled marketing environments.
What a DPDP-Ready Consent Model Looks Like
A DPDP ready consent model treats consent as an operational asset.
Consent is captured with clear purpose definitions. It is mapped to specific channels. It is stored centrally and enforced across systems. Withdrawal is simple and effective.
Marketing execution is designed around consent availability rather than bypassing it.
This approach supports compliant scaling and reduces long term risk.
Why Pharma Marketing Must Rethink Consent as Execution Logic
Consent is no longer a legal document sitting in the background. It is part of execution logic.
Campaign design, segmentation, automation, and personalisation must all respect consent constraints. This requires changes in how teams think about data and engagement.
While this may feel restrictive initially, it ultimately leads to cleaner data, more relevant communication, and stronger trust with doctors.
Frequently Asked Questions on Consent Under DPDP
Closing Perspective and CTA
Pre-DPDP consent models were built for speed and scale, not for accountability. Under the Digital Personal Data Protection Act, these models are no longer sufficient.
Pharma marketing teams must treat consent as a core execution control rather than a background formality. Those that redesign early will be able to operate with confidence and scale responsibly.
If you are assessing how to move from legacy consent practices to DPDP-compliant HCP marketing, this page explains how consent-first doctor engagement is being operationalised in real pharma workflows.
