DPDP Act 2023 Explained For Pharma Marketing Teams

The Digital Personal Data Protection Act, 2023 has quietly but fundamentally changed how pharmaceutical marketing operates in India. While many discussions around DPDP focus on legal interpretation, fines, or policy language, the real impact of this law is being felt much closer to the ground.
It is reshaping how doctor data is collected, how campaigns are executed, how agencies operate, and how accountability is distributed inside pharma organizations.
For pharma marketing teams, DPDP is not a future compliance exercise or a document to be handled only by legal or IT.
It is an operational reality that now governs everyday activities such as maintaining doctor databases, running email and WhatsApp campaigns, deploying CRM systems, and using AI for personalization.
This article explains DPDP Act 2023 in practical terms for pharma marketing teams, without legal jargon and without oversimplification.
Why DPDP Matters Specifically to Pharma Marketing ?
Pharma companies sit in a unique position when it comes to data. Unlike many consumer businesses, pharma marketing involves regulated products, professional audiences, sensitive contexts, and a long history of data being shared across vendors, agencies, and internal systems.
For years, doctor data was treated as a commercial asset. Databases were purchased, enriched, exchanged, segmented, and activated across channels with minimal scrutiny. Consent was often assumed, bundled, or implied through professional interactions.
DPDP changes this foundation.
Under DPDP, most of the data used in pharma marketing qualifies as personal data. This includes doctor names, phone numbers, email IDs, location details, digital identifiers, engagement history, and even inferred preferences when linked back to an individual.
This is why DPDP-compliant HCP marketing is no longer optional but foundational to how pharma engagement must be designed and executed.
What the DPDP Act 2023 Actually Says in Simple Termsa ?
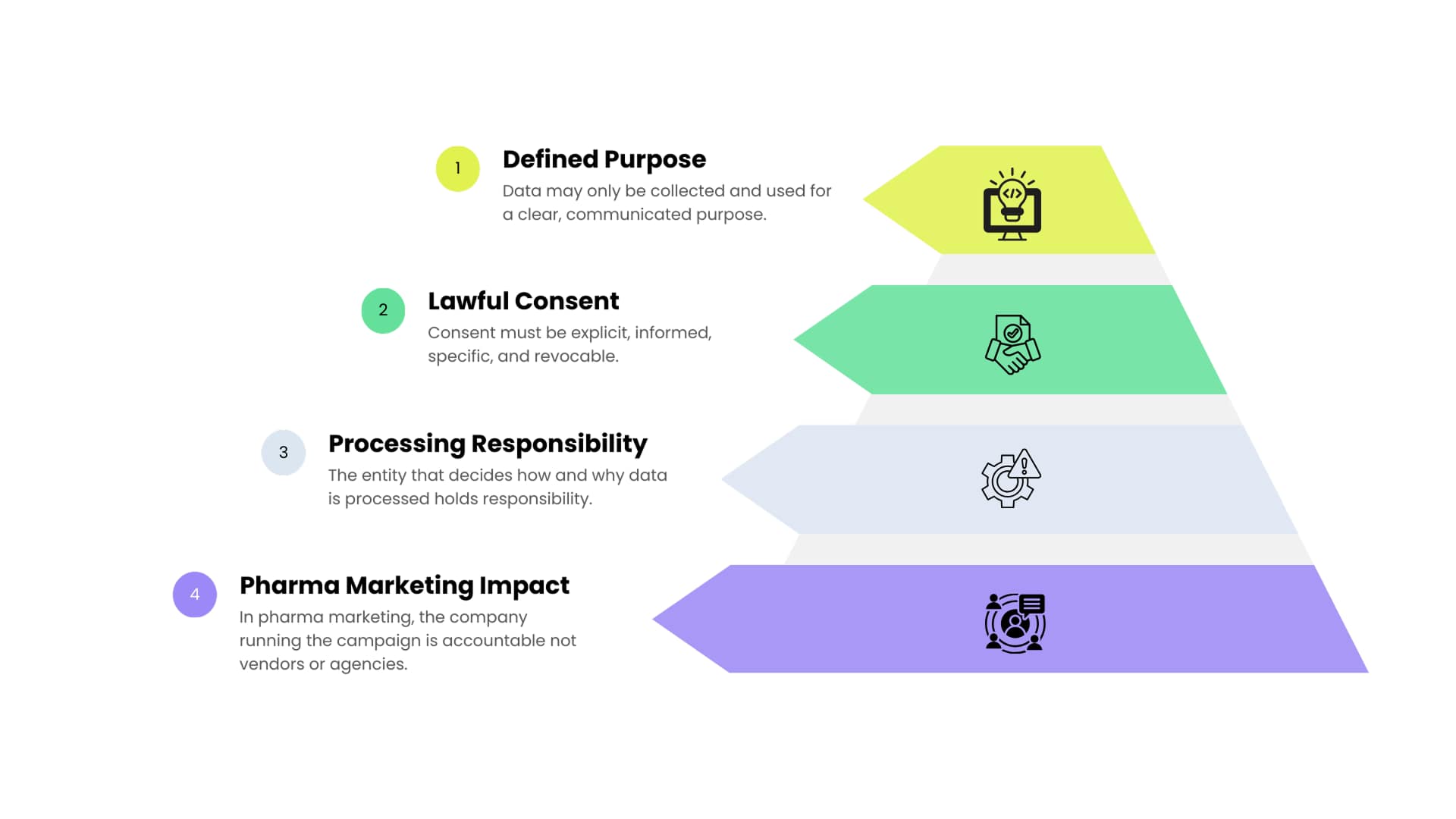 At its core, the DPDP Act governs how digital personal data can be processed. Processing includes collection, storage, sharing, analysis, and use.
At its core, the DPDP Act governs how digital personal data can be processed. Processing includes collection, storage, sharing, analysis, and use.
The law introduces three concepts that matter deeply to marketing teams.
First, the idea of purpose limitation. Data can only be collected and used for a clearly defined purpose that has been communicated to the individual.
Second, consent as the primary legal basis. Consent must be explicit, informed, specific, and capable of being withdrawn.
Third, accountability of the data fiduciary. The entity that decides why and how data is processed is responsible for compliance, regardless of how many vendors or agencies are involved.
For pharma marketing, this means the company running the campaign is accountable, not the CRM vendor, not the digital agency, and not the data supplier.
DPDP Act Full Form and Why It Signals a Structural Shift
DPDP stands for Digital Personal Data Protection Act.
This is not just another data protection guideline or advisory. It is a binding law that signals India’s move toward stricter governance of digital data, closer to global regimes like GDPR, but with its own structure and enforcement philosophy.
What makes DPDP particularly impactful for pharma is that it arrives at a time when marketing execution has become deeply data driven. CRM systems, omnichannel platforms, AI driven segmentation, and automated outreach are now central to commercial success.
DPDP does not ban these practices. But it forces them to mature.
How Pharma Marketing Worked Before DPDP ?
To understand the change, it helps to be honest about how marketing workflows typically functioned earlier.
Doctor databases were often sourced from multiple vendors and merged into a central CRM. Consent, if tracked at all, was usually generic and static. Campaigns were designed around reach, frequency, and segmentation logic rather than consent status. Agencies ran digital campaigns with limited visibility into how data had been sourced or whether permissions were current.
In this model, compliance was largely assumed. The risk was considered low, enforcement was minimal, and accountability was diffused.
DPDP makes this model untenable.
How DPDP Changes Doctor Databases ?
One of the first areas where DPDP has a direct impact is doctor databases.
Under DPDP, it is no longer enough to know that a doctor’s data came from a reputed vendor or a long standing source. What matters is whether the data was collected lawfully, whether consent exists for the current use case, and whether that consent can be demonstrated.
Marketing teams now need answers to questions that were previously ignored. When was consent obtained. For what purpose. Through which channel. Is the consent still valid. Can it be withdrawn easily.
Databases that cannot answer these questions carry real risk.
Consent Under DPDP Is Not the Same as Opt In
One of the most common misunderstandings in pharma marketing today is equating opt in with DPDP compliant consent.
Opt in typically refers to a one time agreement, often bundled into a form or interaction. DPDP requires something more specific. Consent must be explicit, meaning the individual has clearly agreed. It must be informed, meaning they understand how their data will be used. It must be purpose specific, meaning consent for one activity does not automatically apply to others.
For example, consent to receive scientific updates does not automatically cover promotional messaging, surveys, or AI driven personalization.
This distinction has major implications for campaign planning and execution, especially for teams running multi channel doctor engagement programs.
Impact on CRM Systems Used by Pharma Companies
Many pharma CRMs were designed in an era where data governance was secondary to sales enablement. They excel at segmentation, campaign automation, and performance tracking, but often lack native consent intelligence.
DPDP exposes this gap.
A CRM that cannot dynamically check consent status before triggering outreach becomes a liability. A CRM that stores consent as a static field without purpose mapping or withdrawal logic is not sufficient.
This is where modern DPDP compliant HCP marketing workflows become critical, because consent must travel with the data across CRM, campaign tools, and engagement channels.
Omnichannel Marketing Under DPDP
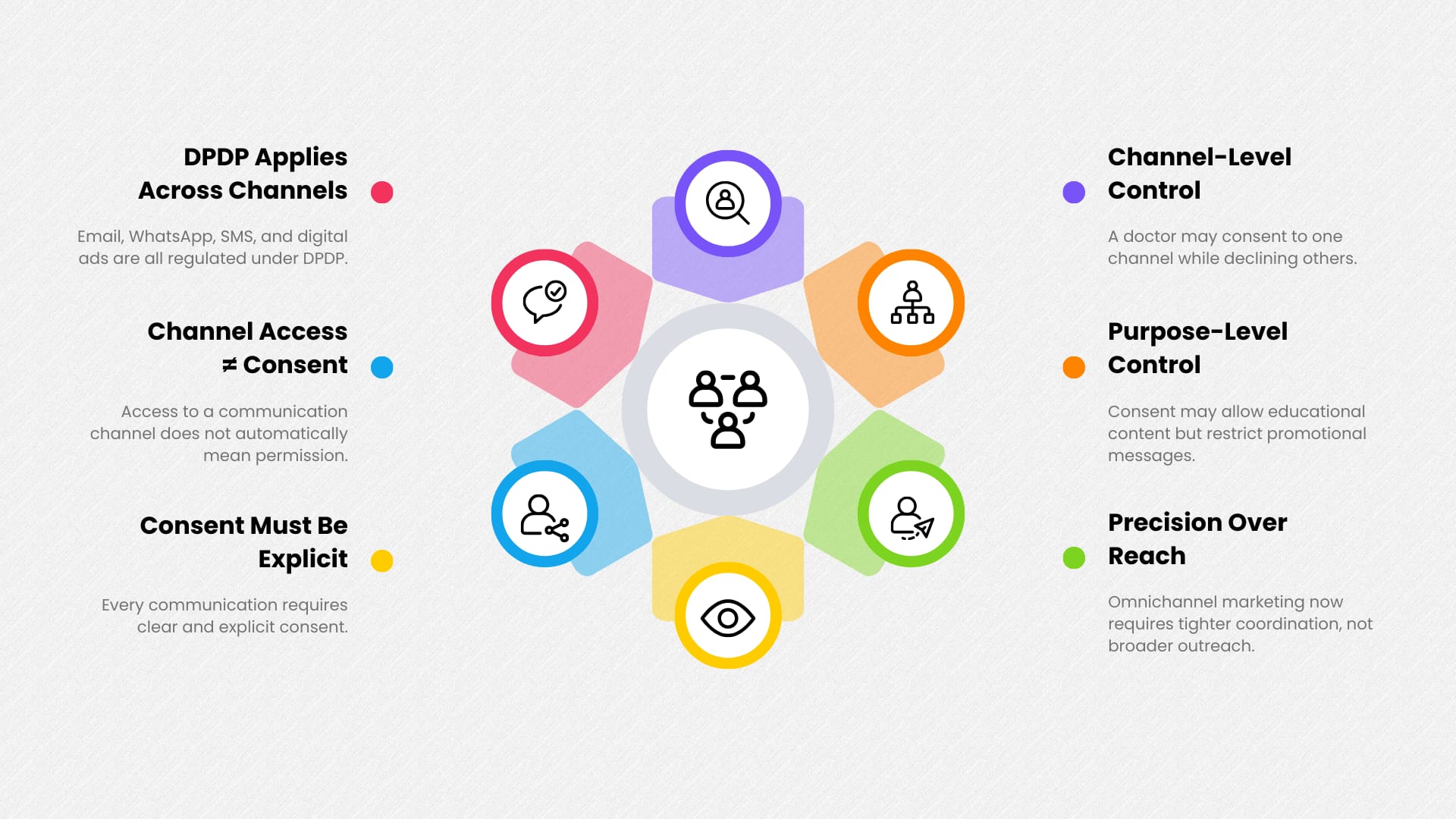
Email, WhatsApp, SMS, and digital ads are all affected by DPDP.
The key shift is that channel access alone is no longer enough. Permission must be tied to both the channel and the purpose.
A doctor may consent to receive email communication but not WhatsApp messages. They may consent to educational content but not brand promotion. DPDP requires these distinctions to be respected in execution.
This means omnichannel marketing needs tighter orchestration, not broader reach.
Role of Agencies and Vendors After DPDP
Another area where confusion persists is agency responsibility.
While agencies execute campaigns, DPDP places primary accountability on the data fiduciary. In most cases, that is the pharma company.
This does not mean agencies are irrelevant. It means pharma companies must ensure that agencies operate within clearly defined, DPDP compliant workflows. Data sharing agreements, consent verification mechanisms, and audit readiness all become essential.
Outsourcing execution does not outsource liability.
DPDP in the Context of AI and Personalization
AI is increasingly central to pharma marketing, from next best action engines to content personalization.
DPDP does not prohibit AI. But it raises the bar for data governance. Training data must be lawful. Outputs must respect the original purpose of data collection. Consent withdrawal must propagate across AI systems, not just databases.
This requires closer alignment between marketing, data, and technology teams.
What DPDP Means for Daily Marketing Decisions ?
At a practical level, DPDP forces marketing teams to slow down slightly and think more deliberately.
Before launching a campaign, teams must ask whether the data being used is consented for this purpose. Before expanding to a new channel, they must consider whether consent covers that channel. Before onboarding a new vendor, they must evaluate data handling practices.
This shift directly affects how HCP marketing programs are designed under DPDP, especially when scale and automation are involved.
Frequently Asked Questions on Purpose Limitation Under DPDP
Closing Perspective and CTA
DPDP Act 2023 is not a legal footnote for pharma marketing teams. It is a structural shift in how doctor engagement must be designed, executed, and governed.
Pharma companies that invest early in consent first data foundations, compliant CRM workflows, and audit ready engagement models will operate with far greater confidence in the coming years.
If you are evaluating how to run doctor engagement and HCP marketing under DPDP without increasing operational risk, this page explains how compliant, execution ready HCP marketing is being implemented in practice.
