Data Protection Bill vs DPDP Act:
What Changed for Healthcare ?

For several years, India’s healthcare and pharmaceutical sector operated in a state of regulatory anticipation. Draft versions of the Personal Data Protection Bill circulated, were revised, withdrawn, and reintroduced in different forms. During this period, many healthcare organisations took a wait-and-watch approach, assuming that final compliance requirements would evolve gradually.
The Digital Personal Data Protection Act, 2023 ended that uncertainty.
The DPDP Act is not just a renamed version of the earlier Data Protection Bill. It represents a decisive shift in how personal data is regulated in India, especially for sectors like healthcare and pharma where sensitive, identifiable data is deeply embedded in daily operations.
This article explains what changed between the earlier Data Protection Bill and the DPDP Act, why those changes matter specifically for healthcare and pharma companies, and how marketing, medical, and digital teams must now operate differently.
What Purpose Limitation Means Under DPDP ?
Purpose limitation under DPDP means that personal data must be collected for a specific, lawful purpose and processed only for that purpose.
Data cannot be reused simply because it is available. Each use must align with the purpose communicated to the data principal at the time of consent.
If the purpose changes, expands, or evolves, new consent may be required.
This principle forces organisations to think carefully about why data is collected and how it will be used over time.
A Brief Context: From Data Protection Bill to DPDP Act
India’s journey toward comprehensive data protection legislation began with the Personal Data Protection Bill. That bill attempted to create a wide-ranging framework covering personal data, sensitive personal data, and critical personal data, with extensive obligations and classifications.
Over time, feedback from industry, regulators, and policy experts highlighted challenges around complexity, implementation feasibility, and enforcement clarity. As a result, the government chose to simplify and restructure the law.
The outcome was the Digital Personal Data Protection Act, 2023.
Rather than being an incremental update, DPDP is a re-architected law that focuses on digital personal data, clearer accountability, and more enforceable obligations.
For healthcare, this simplification does not mean reduced responsibility. In many ways, it increases operational accountability.
Key Structural Differences Between the Two Laws
One of the most important changes is scope.
The earlier Data Protection Bill attempted to regulate all personal data, whether processed digitally or otherwise. DPDP narrows the scope to digital personal data. For healthcare and pharma, this still covers the majority of operational data because patient records, doctor databases, CRMs, analytics platforms, and AI systems are digital by design.
Another change is simplification of data categories. The earlier bill introduced multiple categories such as sensitive and critical personal data. DPDP removes these classifications and instead focuses on personal data with contextual obligations.
For healthcare organisations, this means fewer theoretical distinctions but clearer responsibility for how data is handled in practice.
What Changed in Consent Requirements ?
Consent existed in the earlier Data Protection Bill, but DPDP makes consent far more central and operational.
Under DPDP, consent must be explicit, informed, specific to a purpose, and easy to withdraw. These requirements are clearer and more enforceable than in earlier drafts.
For healthcare and pharma marketing teams, this has immediate consequences. Doctor engagement, patient education programs, digital campaigns, and outreach initiatives must now be designed with consent as a live condition, not a one-time approval.
Earlier models that relied on broad or implied consent are no longer sufficient.
Accountability Shift for Healthcare Organisations
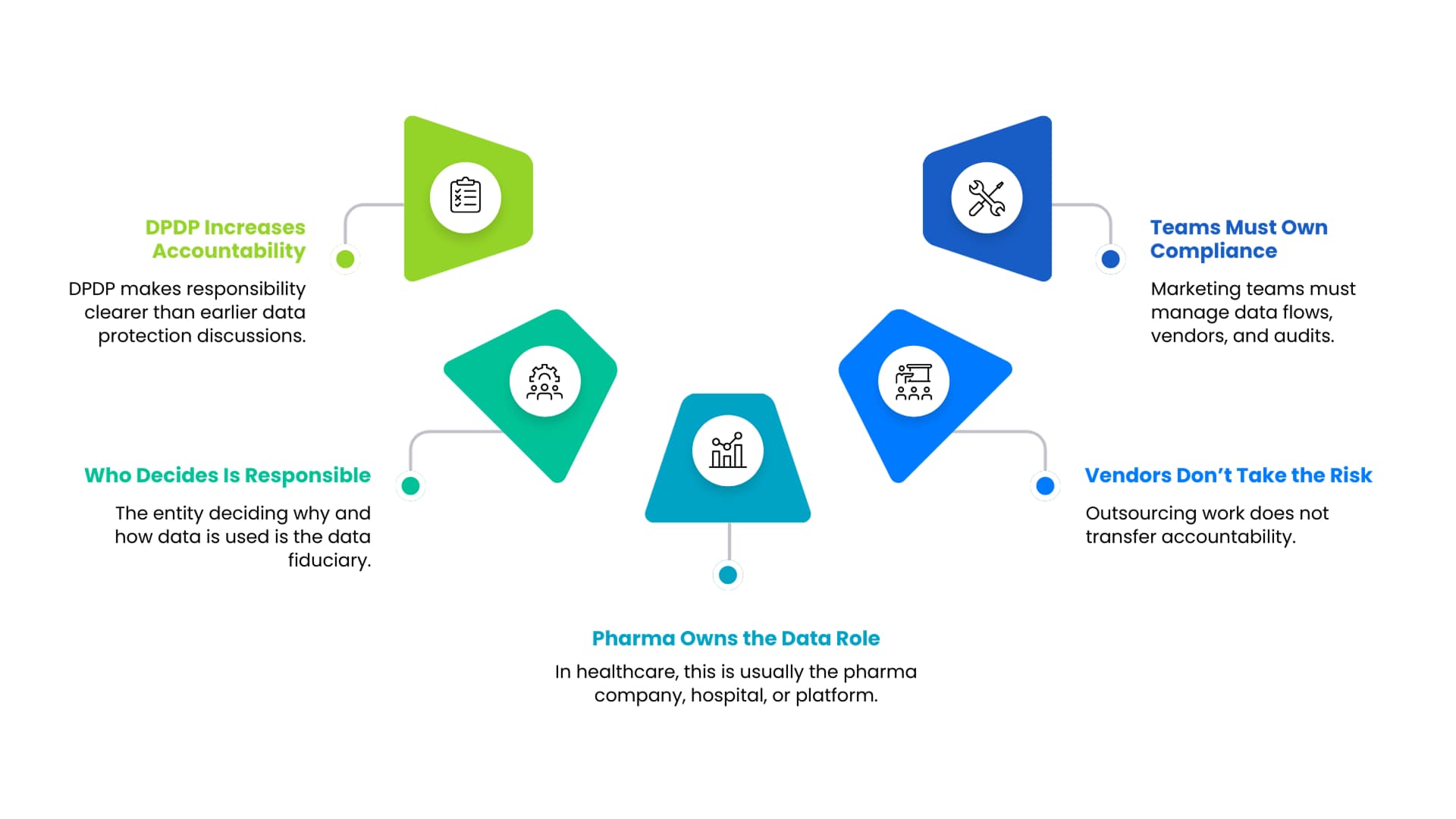
The Data Protection Bill discussed obligations, but DPDP sharpens accountability.
Under DPDP, the entity that determines why and how personal data is processed is the data fiduciary. In healthcare and pharma, this is almost always the hospital, pharma company, or healthcare platform, even when vendors or agencies are involved.
This removes ambiguity. Outsourcing execution does not outsource responsibility.
For healthcare marketing and commercial teams, this means greater ownership of data workflows, vendor governance, and audit readiness.
Impact on Doctor and Patient Data Handling
Under the earlier bill, many healthcare organisations assumed that professional data such as doctor contact information would be treated differently from consumer data.
DPDP does not support this assumption.
Doctor data is personal data if it can identify an individual. Patient data is also personal data, often highly sensitive in nature. The law does not dilute obligations simply because data is used in a professional or clinical context.
This change forces healthcare organisations to reassess long-standing practices around data sharing, enrichment, and reuse.
What Changed for Healthcare Marketing and Engagement ?
Healthcare marketing sits at the intersection of compliance and communication. The DPDP Act affects how hospitals, pharma companies, and health platforms engage both doctors and patients.
Under DPDP, campaigns must align clearly with the purpose for which consent was obtained. Channel specific consent becomes important. Data reuse across campaigns must be justified.
This is why DPDP-compliant HCP marketing has become a core requirement rather than an optional enhancement.
CRM and Digital Systems Under DPDP
Earlier, many healthcare organisations treated CRMs and marketing tools as operational utilities, with compliance layered on externally.
DPDP changes this approach.
Systems must now support consent tracking, purpose mapping, and enforcement at the point of execution. A CRM that cannot prevent outreach when consent is missing introduces risk.
The difference between the Data Protection Bill and DPDP here is practical enforceability. DPDP expects systems to work correctly, not just policies to exist.
Penalties and Enforcement Philosophy
The earlier Data Protection Bill discussed penalties, but enforcement mechanisms were less clear and often perceived as distant.
DPDP introduces clearer penalty structures and a stronger enforcement posture. This does not mean penalties will be applied indiscriminately, but it does mean organisations must be prepared to demonstrate compliance.
For healthcare organisations, reputational risk often outweighs financial penalties. A compliance failure involving patient or doctor data can damage trust quickly.
Why DPDP Leaves Less Room for Interpretation ?
One of the biggest differences healthcare leaders will notice is reduced ambiguity.
The Data Protection Bill allowed room for interpretation and delayed readiness. DPDP is clearer about expectations and rights.
Consent must exist. Purpose must be defined. Withdrawal must be respected. Accountability must be demonstrated.
This clarity accelerates the need for action.
Implications for AI and Data Analytics in Healthcare
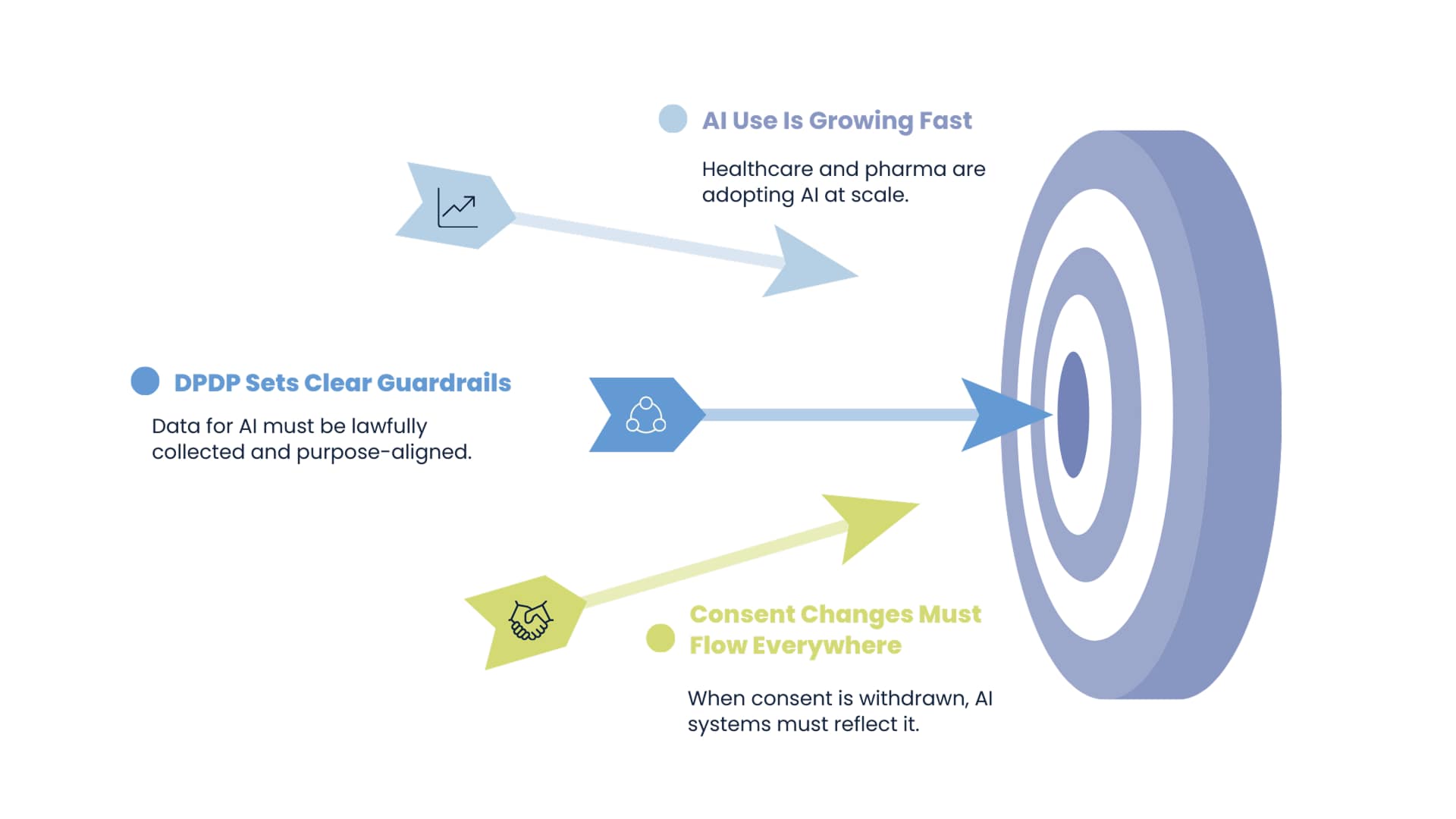
AI adoption in healthcare and pharma has accelerated rapidly. The DPDP Act introduces guardrails that affect how AI models are trained and used.
Data used for AI must be lawfully collected. AI outputs must align with the original purpose of data collection. Consent withdrawal must propagate across systems.
The earlier bill discussed these ideas but did not create strong operational expectations. DPDP does.
This forces healthcare organisations to integrate governance into AI strategy rather than treating it as a separate concern.
Why Healthcare Can No Longer Delay Action ?
During the Data Protection Bill phase, many healthcare organisations delayed major changes, expecting further revisions.
DPDP removes that option.
The law is active. Expectations are defined. Enforcement pathways exist.
Healthcare organisations that continue to operate on legacy assumptions risk operational disruption, audits, and loss of trust.
Frequently Asked Questions on Data Protection Bill vs DPDP Act
Closing Perspective and CTA
The shift from the Data Protection Bill to the Digital Personal Data Protection Act marks a turning point for healthcare and pharma companies.
What was once a future compliance consideration is now a present operational requirement. Healthcare organisations must move from policy level readiness to execution level discipline.
If you are assessing how to operationalise DPDP-compliant HCP marketing and healthcare engagement, this page explains how consent-first, compliant workflows are being implemented in real-world environments.
